The atomic theory

the ion with 2 fewer oxygens than the most common ionic form is called hypo- .the ion with 1 less oxygen than the most common ionic form is called -ite ion.Chemical compounds are made up of at least 2 atoms of. Atoms of different elements vary in size, mass and chemical behavior. All atoms of the same element are identical and have the same mass. Best Known For: Chemist John Dalton is credited with pioneering modern atomic theory. Here are the main points: Matter is made up of atoms, small and indivisible particles.

The most common form of the oxoanions in a series is named by changing the ending of the central element's name to -ate and adding the word ion The atomic theory was formulated by John Dalton. Explore the key discoveries and models of atoms, such as Democritus' atomism, Dalton's law of multiple proportions, Rutherford's plum pudding model, Bohr's model, and quantum mechanics. Electrons are attracted to the protons in the. Learn how atomic theory evolved from ancient philosophy to modern quantum mechanics. Protons and neutrons form the atomic nucleus. The three parts of the atom are protons (positively charged), neutrons (neutral charge), and electrons (negatively charged). atomic model, in physics, a model used to describe the structure and makeup of an atom. They often occur in series of 2 or more ions with the same central atom but different numbers of oxygens An atom is a building block of matter that cannot be broken apart using any chemical means. Polyatomic anions that contain one or more oxygen atoms and one central atom of another element. An atom is the smallest unit of an element that can participate in a chemical change. Matter is composed of exceedingly small particles called atoms. Here are the postulates of Dalton’s atomic theory. named by changing the ending of the element's name to -ide and adding the word ion First published in 1807, many of Dalton’s hypotheses about the microscopic features of matter are still valid in modern atomic theory.if there are more than one possible charge (especially for the transition metals), a Roman numeral is added behind the name of the elements to indicate the charge Nuclear power is considered one of the ways to reduce dependence on fossil fuels, but how to deal with nuclear waste products is a concern. Atom, smallest unit into which matter can be divided without the release of electrically charged particles.named by adding the word ion to the name of the elements.Matter is made up of atoms that are indivisible and indestructible. The law of conservation of mass states that the total mass present before a chemical reaction is the same as the total mass present after the chemical reaction. Modern Atomic Theory (John Dalton) Experiments with gases that first became possible at the turn of the nineteenth century led John Dalton in 1803 to propose a modern theory of the atom based on the following assumptions. Scientists did not account for the gases that play a critical role in this reaction.
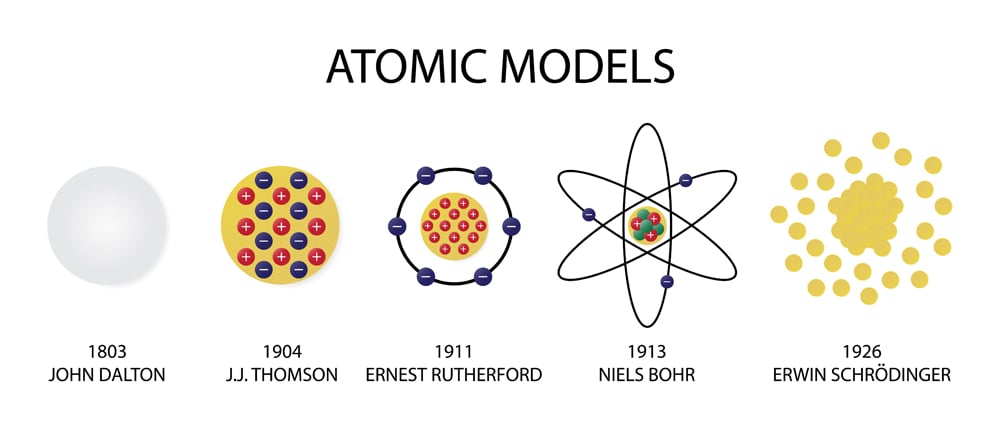
The first part of his theory states that all matter is made of atoms, which are indivisible. Dalton based his theory on the law of conservation of mass and the law of constant composition. \) shows that the burning of word does follow the law of conservation of mass. Dalton's atomic theory was the first complete attempt to describe all matter in terms of atoms and their properties.
